Introduction
SDTMIG: The Study Data Tabulation Model Implementation Guide (SDTMIG) for human clinical trials guides users on the organization, structure, and format of standard clinical study tabulation datasets for interchange between organizations or to be submitted to a regulatory authority. The following videos introduce you to the SDTMIG and the SDTM, which used together serve as a map that orients you on how your data fits into the standard.
Versions
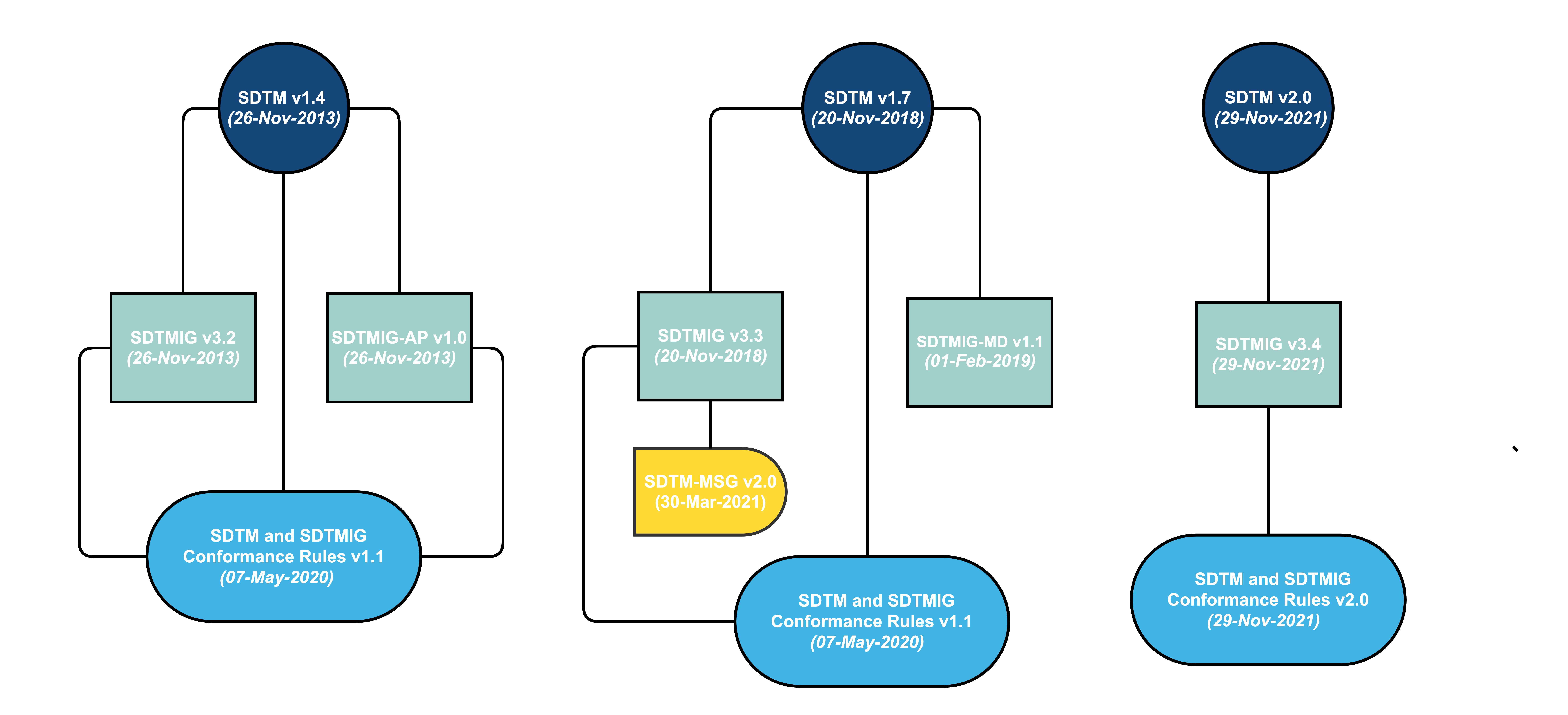
A Study Data Tabulation Model Implementation Guide (SDTMIG) is developed in reference to a specific SDTM model. However, the SDTM is cumulative – each new release builds on the previous model. Therefore, the models are backward compatible. For example, SDTMIG-AP v1.0 was developed in reference to SDTM v1.4, but it may be used in a submission that uses SDTM v1.7.
Implementers should be aware that if they are referencing a model for which the IG was not originally developed, variables may have been added or deprecated from the model. In addition to models and implementation guides, conformance rules have been developed, which help to ensure that generated data structures conform to the standards. These rules aim to identify all conformance rules and case logic from the SDTM and SDTMIG, classifying and codifying them in a form that supports quality processes and tool development.
Therapeutic Area User Guides
Therapeutic Area User Guides (TAUGs) extend the Foundational Standards to represent data that pertains to specific disease areas. TAUGs include disease-specific metadata, examples and guidance on implementing CDISC standards for a variety of uses, including global regulatory submissions. The following video will introduce you to TAUGs and how they relate to CDISC Foundational Standards.
Controlled Terminology
Controlled Terminology is the set of codelists and valid values used with data items within CDISC-defined datasets. The following video introduces you to CDISC Controlled Terminology and how it is used with CDISC standards.
Traceability
Traceability is a fundamental element of data quality and a requirement for studies submitted to regulatory authorities. From data collection to final analysis, traceability plays a crucial role in ensuring the integrity of source data and in reinforcing clinical research results. The following video provides an introduction to implementing traceability in CDISC-compliant studies.
Regulatory Requirements
CDISC standards are required or recommended by several global regulatory agencies. Standardized data enables regulators to streamline the review process with a more consistent use of analysis tools to better view drug data and highlight areas of concern. The following video introduces you to regulatory requirements and the use of CDISC standards.
Team Guiding Principles
Standards in Development
Foundational
For current versions of the standards, please visit the Standards Home Page.
| Standard Sort descending | Release Notes | Projected Publication |
|---|---|---|
| ADaM and IG v3.0 | In Development |
2026 |
| CDASH v2.0 | In Development |
2027 |
| CDASHIG v3.0 | In Development |
2026 |
| SDTM v3.0 | Resolving Public Review comments. |
2026 |
| SDTMIG v4.0 | Resolving Public Review Comments |
2027 |
| SENDIG v4.0 | Resolving Public Review Comments |
2026 |
Data Exchange
For current versions of the Data Exchange standards, please visit the Data Exchange Page.
| Standard Sort descending | Release Notes | Projected Publication |
|---|---|---|
| Controlled Terminology Package 62 | In Development |
2026 |