Latest page content update: 15 May 2026

Introduction
CDISC Conformance Rules are an integral part of the Foundational Standards and provide specific guidance to industry for the correct implementation of the standards.
Today, these rules:
- Are published in Excel format and are not machine-executable
- Can be interpreted and implemented differently across organizations
- Could lead to inconsistencies in data validation and regulatory submissions
As industry best practice shifts towards applying Conformance Rules throughout the study lifecycle, to keep data as submission ready as possible, the need for a unified, reliable approach has become essential.
The CDISC Open Rules Project directly addresses this need by providing an aligned, transparent, and machine-executable rules framework that supports consistency from study design through regulatory submission.
Project Goals and Objectives
The overall goal of the CDISC Open Rules Project is:
- To deliver a governed set of unambiguous and executable Conformance Rules (CDISC Open Rules) for each Foundational Standard
- To provide a Reference Implementation of an open-source execution engine (CDISC Open Rules Engine, abbreviated as CORE) for the CDISC Open Rules.
The objectives of the CDISC Open Rules Project are:
CDISC Open Rules
- Ensure each standard has a set of unambiguous, machine-executable Conformance Rules
- Create these CDISC Open Rules vetted by the CDISC standards development teams
- Ensure consistency across Conformance Rule implementations
- Expedite the availability of CDISC Open Rules for new Foundational Standards
CDISC Open Rules Engine (CORE)
- Create a Reference Implementation of an open-source engine that executes the CDISC Open Rules
- Release the open-source engine under the CDISC Open-Source Alliance (COSA)
Project Concept
The following diagram illustrates the concept of the project, including the Conformance Rules, the machine-executable form of the rules (CDISC Open Rules), and the Rules execution engine (CORE):
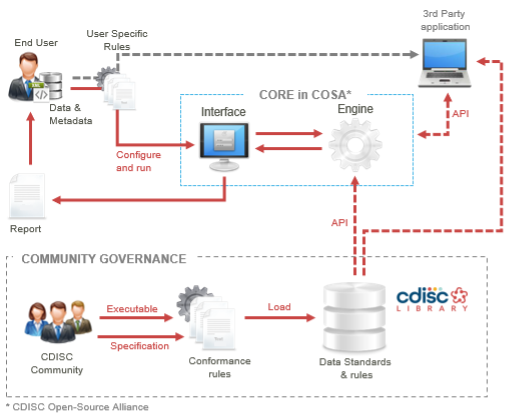
CDISC Open Rules
The CDISC Open Rules are expressed in a simple, low code format (YAML) supported by a clear reference structure, enabling easy, transparent, and consistent rule creation, review, and testing.
CORE
The open-source engine is built to execute the rules reliably and repeatably across datasets. The CDISC Open Rules Engine (CORE), is available on GitHub: cdisc-rules-engine
The research community can leverage this free, open-source solution to test study data for conformance to CDISC standards, as well as regulatory and sponsor-specific rule sets (= custom rules).
Considerations
- The CDISC Open Rules Project includes development of:
- Executable Conformance Rules for the CDISC standards
- Executable Conformance Rules for FDA Business Rules
- A Reference Implementation of a software engine (CORE) to execute these rules
- CDISC is publishing the executable Conformance Rules in the CDISC Library
- CDISC provides free access to CORE to CDISC members and non-members
- CORE is published as open source (MIT license)
- CDISC has no plans to deploy CORE as commercial software
- Developers have the option to prepare CORE for:
- On-premises deployment
- A cloud-platform deployment
- Running from command line or package, integrating with other systems
- Implementers may choose to develop a proprietary engine or adapt CORE. The CORE Reference Implementation can be used to confirm that a proprietary engine can execute the CDISC Open Rules with the correct results
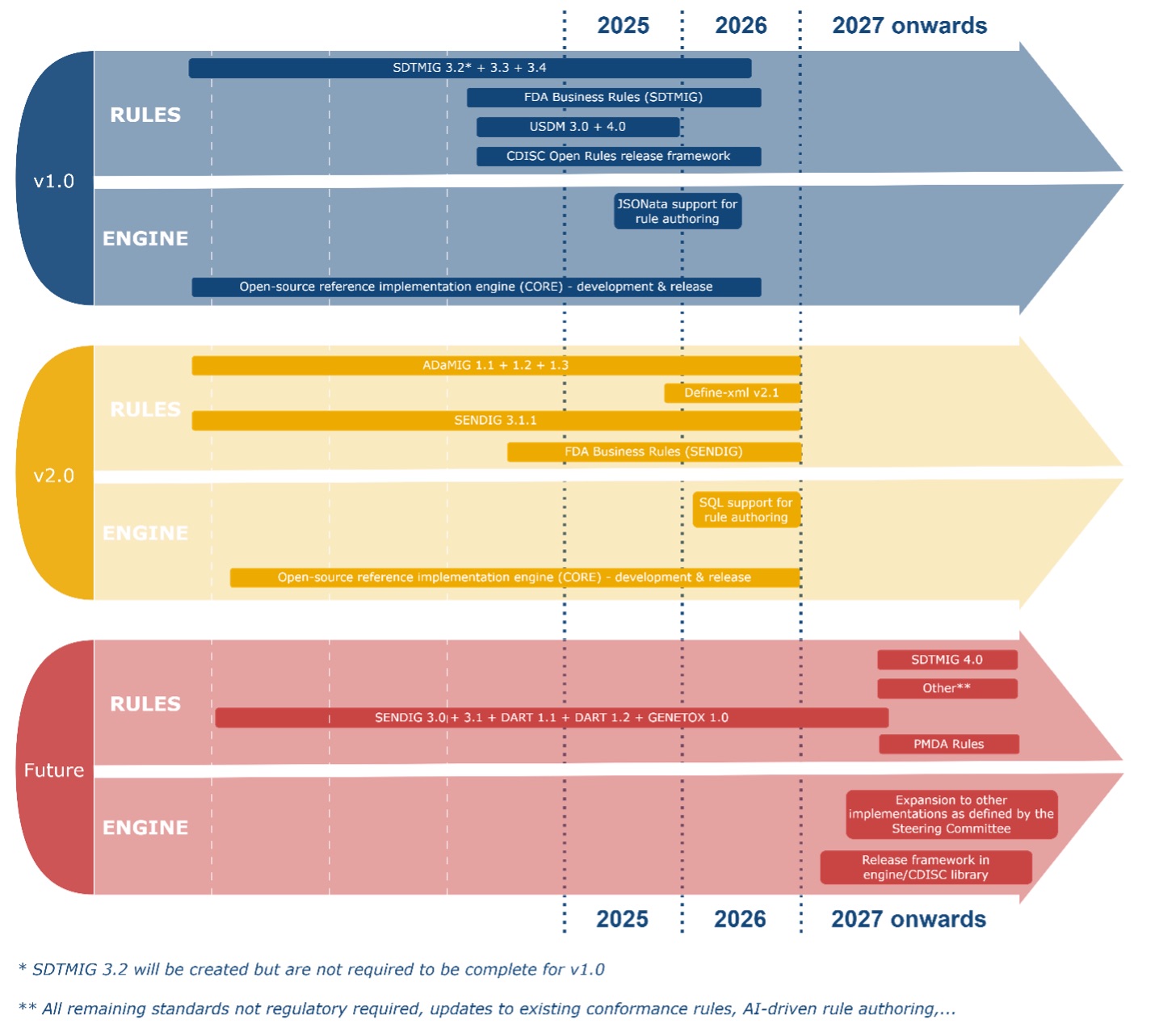
CDISC Open Rules Roadmap
Note that Roadmap timelines are estimates, depend on volunteer‑driven progress, and will be updated regularly to reflect the latest projections.
How to Participate
Join us in advancing the CDISC Open Rules Project. You and your organization’s participation can help drive meaningful industry progress.
Visit the CORE Project Participate tab to learn how you can contribute.
Additional Resources
CDISC Announces Research Collaboration with FDA to Incorporate FDA Business Rules into CORE
Latest page content update: 15 May 2026
Next Steps
- Complete the remaining Open Rules as per Roadmap targets.
- Transition rule authoring from the Rule Editor to a GitHub-based workflow, supporting higher-quality and more scalable rule development.
- Implement a formal governance process for Conformance Rules.
- Continue dialog with software vendors considering development of CORE offerings.
- Implement a certification program for proprietary CDISC Open Rules execution engines.
- Support formal governance via the Steering Committee, with industry and regulatory representation.
- Finalize engine refactoring, merging, and release of CORE V1.0 (targeted mid-2026).
Milestones
2021
- MVP Development Begins
Initial development of the MVP CDISC Open Rules Editor and Engine, laying the foundation for executable rule creation. - First Volunteer Rule Authoring
Early access to the MVP tools enabled volunteers to begin authoring conformance rules, starting with SDTMIG 3.4.
2022
- MVP Engine Released
The first MVP CORE Engine was launched in the CDISC cloud at the CDISC EU Interchange. Rule development accelerated with two-thirds of SDTMIG 3.4 executable rules published and available through the CDISC Library API. - Transition to Open Source
The CORE Engine and Rule Editor moved to an MIT license, COSA-governed open-source model published on GitHub with a CLI, supported by a new Roadmap Board and expanded volunteer training.
2023
- First Desktop Deployment Demonstration
An industry provider showcased the first desktop deployment concept for the CORE Engine, helping illustrate how users could run the engine locally with minimal setup.
2024
- FDA Collaboration Established
A research collaboration agreement was signed with the FDA to author FDA Business Rules in the CORE syntax, alongside continued discussions with industry on system integration. - First Company-Specific Rules Authored
Organizations began creating custom company-specific rules, demonstrating practical use of the CORE syntax across internal workflows and lifecycle processes. Examples were showcased during the COSA webinar.
2025
- CDISC Open Rules Accelerator Program Launched
CDISC invited organizations to join the Accelerator Program, contributing resources or funding to speed rule development. Participants receive governance roles and recognition. - CORE Refactoring Initiated to Accelerate CDISC Open Rules Development
CDISC partnered with Verisian to refactor the CORE Engine to improve performance, flexibility, and usability, accelerating the creation and adoption of CDISC Open Rules.
2026
- 83% of Target Rule Set Completed
By early 2026, authors completed 83% of the planned rule set (1,563 of 1,880 rules) across SDTMIG, SENDIG, FDA Business Rules, TIG, and USDM. - Migration to GitHub Workflow & Planned Engine Merge
Rule authoring fully migrated to a GitHub-based workflow, improving transparency and scalability. A merge of CDISC’s reference engine with the Verisian SQL-based engine is planned for mid-2026.
Latest page content update: 15 May 2026
Project milestone reached: 28 April 2022
Evaluation version of CORE Engine Reference implementation was deployed on CDISC’s Azure cloud subscription:
- Available to users for free
- Initially includes 200+ Rules for SDTMIG v3.4
- Test data is provided by CDISC; users cannot provide their own data
- Visit the TRY IT NOW tab to access this deployment
Project milestone reached: 31 August 2022
Transition of the CORE Engine Reference Implementation to the open-source environment was completed with provision of the Engine in GitHub. The Engine is:
- Provided as open-source with the permissive MIT license
- Registered with the CDISC Open-Source Alliance (COSA)
- Available to users for free
- Provided with a command line interface (CLI)
- Accessed at the GitHub CDISC-rules-engine repository, including special instructions in the Readme file
CORE Rules Development
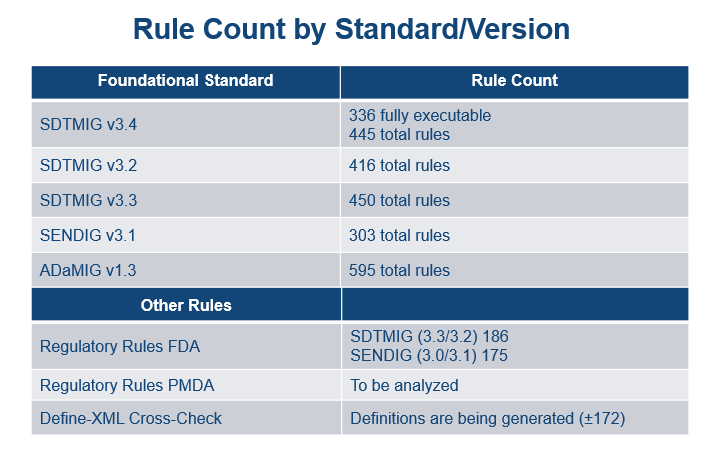
Rules Planning
- An inventory of Rules per Foundational Standard has been developed and is presented in the following graphic:
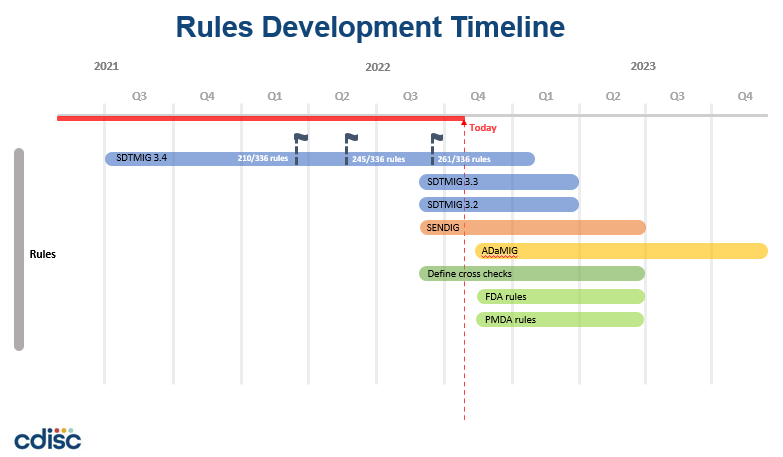
- A timeline for development for these Rules has been prepared and is presented in the following graphic. Note that much of the work for SDTM 3.4 has been completed already.
- The remaining work represented in the timeline is estimated to require about 3.0 FTE for about 8 months, followed by about 1.0 FTE for an additional 6 months. This timeline assumes this level of resourcing will become available to the project.
Rules Status
- The work on the SDTM, SEND, and Define Cross-Checks has recently begun with the limited number of volunteers currently available.
- The CORE project is actively recruiting additional volunteers for this work right now. (See the CORE Roadmap Board activities below on this web page (tab) for more about this).
- A “CORE Volunteer Onboarding Training” webinar for this Rules development was held June 7, 2022. The webinar covered the scope of Rules, the Rule Editor for authoring and testing Rules, and how to sign up and participate. Listen to the CORE Volunteer Onboarding Training. We invite you to visit the Participate tab to sign up to participate on CORE.
- Beyond the webinar, other on-boarding materials to support Volunteers have been delivered:
- Training materials
- Quarterly training sessions to onboard new rule authors
- Rules development workshops at:
- 2022 CDISC US Interchange
- 2022 PHUSE EU Connect
CORE Engine Development
Engine and Deployments Planning and Status
- The evaluation version of the CORE Engine Reference Implementation was previously deployed to the CDISC cloud subscription (April 2022) and to the Azure Marketplace (June 2022).
- Following these deployments, and as part of the transition of CORE from a traditional CDISC project to an open-source framework, CDISC has released the Engine on GitHub.
- Going forward, the open-source Engine development community will be responsible for future Builds of the Engine. These Builds will continue to improve functionality and features of the Engine, as needed. The team anticipates multiple Beta releases with less frequent Stable releases made available. All Engine Builds will be made available on GitHub.
- Engine deployments will be the responsibility of the users. This will include end-users such as Pharma-Biotech and CRO organizations as well as commercial software vendors and integrators deploying for end-users.
- Currently, several vendors have confirmed they will publish free desktop deployments. More communication is expected to follow.
- In addition, the CORE project anticipates that a small number of end-users will integrate the Reference Implementation into their environment during 2023.
- The following graphic illustrates this planning:
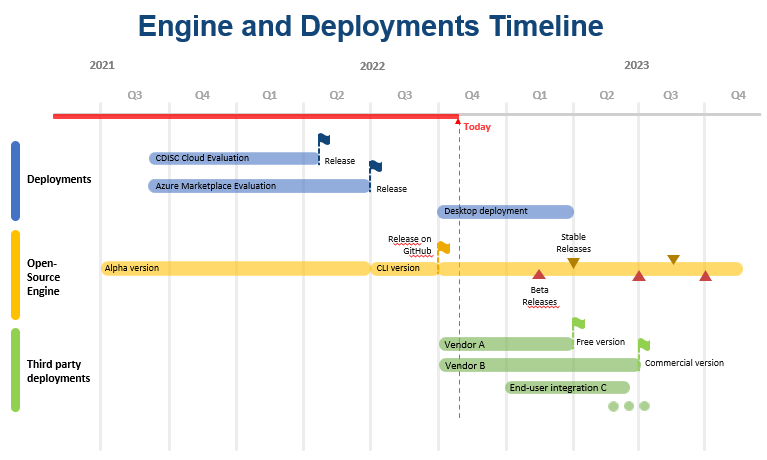
Latest page content update: 24 Nov 2022
Open-Source Governance and Development
During the summer of 2022, after completion of the initial set of executable Conformance Rules (for SDTM 3.4) and release of the evaluation version of the CORE Engine Reference Implementation, CDISC transitioned the CORE project from a traditional CDISC-led project to an open-source framework. The transition focuses on:
- Establishment of a CORE Roadmap Board and CORE Technical Committee to guide and coordinate further Engine Reference Implementation development as open-source and to coordinate with CDISC on CDISC’s further Conformance Rules development.
- Establishment of the CORE Engine Reference Implementation software as open-source by making it publicly available in GitHub and registering it with COSA (CDISC Open-source Software Alliance). Further Engine Reference Implementation development will be conducted under the MIT open-source license by a volunteer, open-source development team.
The following graphic illustrates the full open-source framework for further CORE development. The framework centers on the CORE Roadmap Board and CORE Technical Committee and involves CDISC staff, industry volunteers, and commercial software vendors.
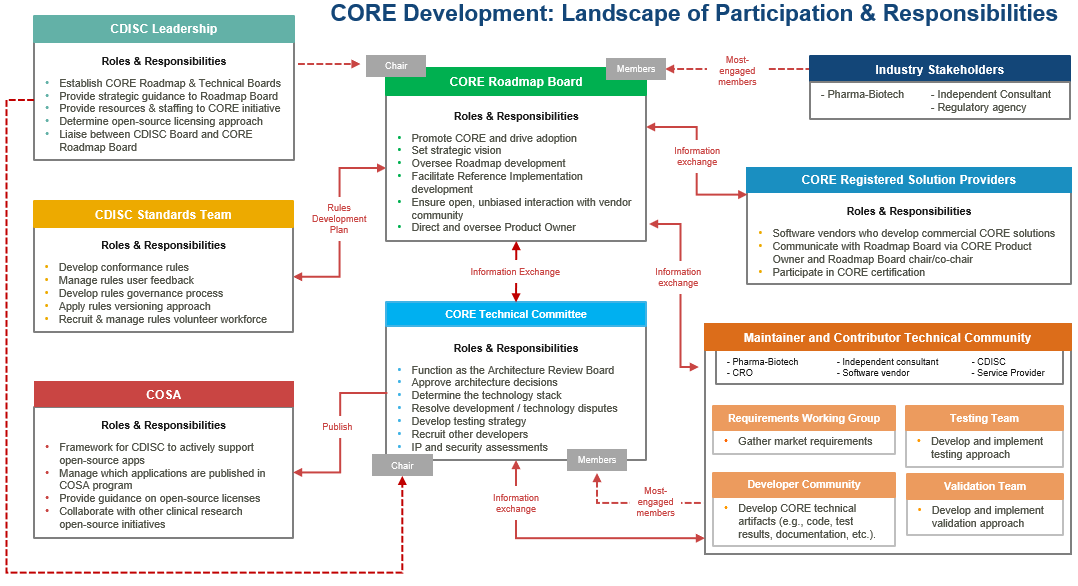
CORE Roadmap Board
The Board is composed of most-engaged CORE stakeholders from Pharma-Biotech, CDISC, Independent consultants, and Service providers. Engaged software vendors have a parallel opportunity to participate today on the CORE Technical Committee and a future opportunity to participate in the CORE Registered Solution Providers program which will be established by the Board.
Following are high-level highlights of the CORE Roadmap Board’s responsibilities:
- Promote CORE and drive adoption
- Set strategic vision
- Oversee Roadmap development
- Facilitate Engine Reference Implementation development
- Ensure open, unbiased interaction with vendor community
Following are the CORE Roadmap Board members:
| Industry | CDISC |
|---|---|
| Anne-Sophie Bekx, Janssen | Dave Evans, CEO |
| Chris Decker, Instem | Sam Hume, VP Data Science |
| Praveen Garg, Astra Zeneca | Peter Van Reusel, CSO |
| Brooke Hinkson, Merck | |
| Stephen Matteson, Pfizer | |
| Girish Rajeev, Takeda |
Board meetings are currently ongoing approximately monthly.
CORE Technical Committee
The Technical Committee is comprised of the most-engaged CORE stakeholders from the CORE Maintainer and Contributor Technical Community. These are the volunteers who are currently most active in the technical side of development of the CORE Engine Reference Implementation.
Following are high-level highlights of the CORE Technical Committee’s responsibilities:
- Function as the Architecture Review Board
- Approve architecture decisions
- Determine the technology stack
- Resolve development / technology disputes
- Develop testing strategy
- Recruit other developers
- IP and security assessments
Latest page content update: 15 May 2026
Join the CDISC Open Rules Community: Accelerator Program and Individual Volunteers
- Mission: Author CDISC Open Rules based on rule specifications and perform Unit Testing to ensure quality and accuracy.
- Work Areas:
- Specification structure and content
- Executable rules content
- Volunteer Skill Set:
Core Skills
- Data-minded with a scientific or analytical background, such as statistics, biometrics, clinical data management, or data science.
- Hands-on experience with CDISC standards, including SEND, SDTM, Define-XML, and/or ADaM.
- Experience working with data specifications and carrying out verification and validation activities.
- Willingness to work in a GitHub + VSCode environment (training provided).
- "Plus” Skills
- Some familiarity with CDISC conformance rules or related rule-based validation processes.
- Experience with structured data formats such as XML, JSON, or YAML.
- Prior experience with Git, GitHub and VSCode
- Participation in an organizational standards council or data governance body.
As the CDISC Open Rules are expressed in a simple, low-code format (YAML) supported by a clear reference structure, people without previous coding experience can be successful in rule development. Our structured onboarding program, combined with ongoing mentoring and community support, ensures that every new volunteer can confidently collaborate and contribute.
CDISC Open Rules Accelerator Program
To accelerate completion of conformance rules across all standards, CDISC has launched the CDISC Open Rules Accelerator Program. The program invites organizations to make a formal, company-level commitment to the project—either through dedicated FTE resources, financial sponsorship, or both.
Joining the Accelerator Program also includes additional benefits and a seat on the Steering Committee.
To apply, please complete the Accelerator Program Interest Form: CDISC Open Rules – Accelerator Program Interest Form. After submitting the form, CDISC will invite your company to an informational call, where you will receive more details about the program and next steps.
Expected Engagement
- Time Period: 6 months commitment, or longer if able
- Hours per week: minimum 20% FTE requested
- Regular meetings and workshops: Recorded and available for review
Sign-up Instructions
- Organizations interested in a formal company-level commitment should apply to the CDISC Open Rules Accelerator Program (see above).
- Individual contributors can sign up on the CDISC volunteer page and select CORE Rules on the form as the Standards Development team. Please include which CORE team you would like to join in the box at the bottom of the page.
CDISC held a webinar 20 July 2021 to provide a deeper understanding of the CORE project, share the “ask” of participants and answer questions from attendees. We invite you to listen to the webinar recording to learn more.
For presentations on CDISC Open Rules and CORE, please visit the CDISC presentations website and COSA channels.
Latest page content update: 15 Mar 2026
Library Access
Please visit the Access tab to set up an account.
- Before CORE was deployed, CDISC has implemented a version of the Library accessible to non-members to ensure that the access needed to use CORE is available to all.
- A CDISC Library API key, accessible through a Library account, will be required for CORE to retrieve executable rules and standards.
Data Format
- CORE ingests a variety of dataset formats, including SAS v5 XPORT, Dataset-JSON, and CSV.
- As an open-source application, vendors can extend CORE to add support for additional dataset formats.
Executable Rules Language and Rules Editor
Most executable rules for CORE (“CDISC Open Rules”) are expressed in YAML, a non-proprietary, human-readable data interchange language.
Rules are authored and managed via GitHub, where contributors collaborate using pull requests and peer review.
Prior knowledge of programming languages is not required to author Rules.
All Rules are based on a shared data model and schema that provides syntax guidance and validation.
USDM conformance rules are expressed in JSONata, a non-proprietary, lightweight query and transformation language for JSON data. JSONata rules follow the same open governance model as YAML-based rules and are stored in the CDISC Library, accessible via the CORE Engine and CDISC Library API.
CDISC maintains rule sets along with publication of foundational standards. Many conformance rules in these rule sets can easily be transcribed into metadata and rule logic, whose structure is bound to the schema. Schema validated rules are sent to CORE for execution.
The Engine provides a framework to plug-in new rule types developed by the CORE team or by other implementers that have the need for additional types of rules.
CDISC plans to only test and validate the rules with their reference engine; however, vendors and sponsors have the option to develop their own applications and custom rules.
Format Reports
- CORE reports will be produced in Excel to facilitate day-to-day operations by CORE users. JSON outputs are also available.
- Vendors and sponsors will have the ability to extend the open-source CORE engine to provide reports in additional output format.
Integration
- The executable Conformance Rules will be available in the CDISC Library, and a rich set of APIs will be provided for the CORE engine to access these rules.
- The open-source CORE engine can be integrated into or run by existing clinical data systems to add a standards conformance capability.
- The CORE system will also provide an API to enable integration with existing clinical data systems.
- In other words, users can create hard endpoint integrations or API endpoints for the CORE engine with their existing systems.
- CDISC plans to create implementation documentation, more information can already be found in the README on GitHub.
- CDISC also has dedicated LinkedIn channel CDISC CORE Collaboration for users to share their experience and integration stories.
Relationship to Commercial Systems
- The CORE engine will be open-source, available to all. It will not be offered by CDISC as a commercial product.
- The CORE engine will be designed to execute the CDISC Conformance Rule sets.
- The CDISC Conformance Rules will be an integral part of the CDISC Standards; they will be developed by CDISC with the Community and will be governed by CDISC standards development processes.
- The CDISC Conformance Rules will be an unambiguous, executable set of rules for CDISC Standards.
- The CDISC Conformance Rules will be a single source of truth, freely available to all in the CDISC Library.
- The CDISC Library will provide a rich set of APIs to access the CDISC Conformance Rules.
- The CDISC Conformance Rules will be expressed in a common/layman language.
- Vendors and sponsors will be able to integrate the open-source CORE engine into their operational systems.
Regulatory Agency
- CDISC works with Regulatory Agencies to ensure input for the conformance rules is compliant with published requirements for each Regulatory Agency. Regulatory Agencies continue to participate in these discussions.
- Yes, CDISC expects to include Regulatory Agency-specific rules within its full set of rules in CORE.
- It is expected that the user can run this specific rule set on the data.
- There are small but important implementation differences between the CDISC Conformance Rules and the Regulatory Agency-specific rules.
- While CDISC continues to work with the Regulatory Agencies to minimize implementation differences, CDISC expects that the user will be able to minimize any differences by checking conformance against multiple rulesets Further, the user will be able to configure the rule sets to meet their needs.
Access to CORE Source Code via GitHub
The CORE source code has been released as open-source on GitHub.
Yes, CDISC will provide training on GitHub, rules creation and running the rules with the command line interface (CLI).
CORE API
Yes, CORE has a REST API.
Validation of a CORE Implementation
CORE will include a suite of automated tests that can be used to help confirm that a CORE implementation is functioning correctly. CDISC also plans to implement a certification program to verify that software that executes the Conformance Rules functions correctly and consistent with the CORE Reference Implementation.
CDISC plans to implement a certification program to verify that any software that executes the Conformance Rules functions correctly and consistent with the CORE Reference Implementation. All CORE Certified software engines will demonstrate that they yield the same results given the same set of rules and CDISC datasets.
Miscellaneous
Going forward, the development cycle for the CDISC Open Rules will become a normal part of the development of standards.
- The CORE engine will be designed to run conformance rules from the CDISC Library, as well as sponsor-defined rules. Sponsor-defined rules will not be maintained in the CDISC Library.
- CORE plans to include Conformance Rule authoring tools that support the development and testing of both CDISC and sponsor-defined rules.
Latest page content update: 15 May 2026
CORE Engine Reference Implementation on GitHub
The CORE Engine Reference Implementation is the current version of the Engine. The CORE Engine Reference Implementation has been transitioned to the open-source environment with its provision on GitHub. The GitHub-based Engine is:
- Provided as open source with the permissive MIT license
- Registered with the CDISC Open-Source Alliance (COSA)
- Available to users for free
- Provided with a command line interface (CLI)
- Accessed at the GitHub CDISC-rules-engine repository, including special instructions in the Readme file
The Engine Reference Implementation on GitHub is an advancement over the CORE Minimum Viable Product (MVP) early evaluation version that was previously available via the CORE web page. In particular, the GitHub-based Engine is updated to process additional Rule syntax (YAML) engaged to handle new processing conditions that were encountered as the CORE team developed additional Rules. The GitHub-based Engine is provided with a command line interface (CLI) and not with a Graphic User Interface (UI).