Links
Published User Guides
TerminologySDTM
Articles
All Public Reviews
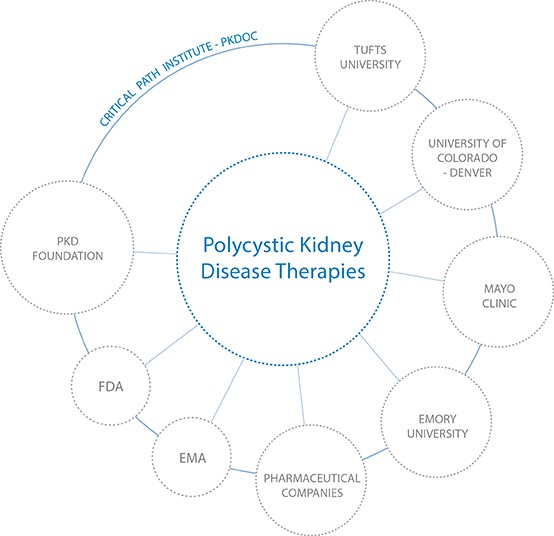
Application of CDISC Standards has supported the discovery of clinical research biomarkers for polycystic kidney disease —the evidence for which had been hidden in non-standardized datasets for years prior to their standardization, aggregation, and analysis. These biomarkers now enable researchers to identify diseases early, when intervention may be possible.