This example shows findings from assessments of step count in a clinical trial in support of DiMe endpoint 399 based on "steps per day"
This example shows findings from assessments of steps taken from an actigraphy device with the purpose of supporting DIME Endpoint 399, "Steps per day," in the regulatory submission of a clinical trial. The submission is for approval of a drug treatment, rather than for approval of the continuous glucose monitor. The CGM data of interest are defined in the study protocol.
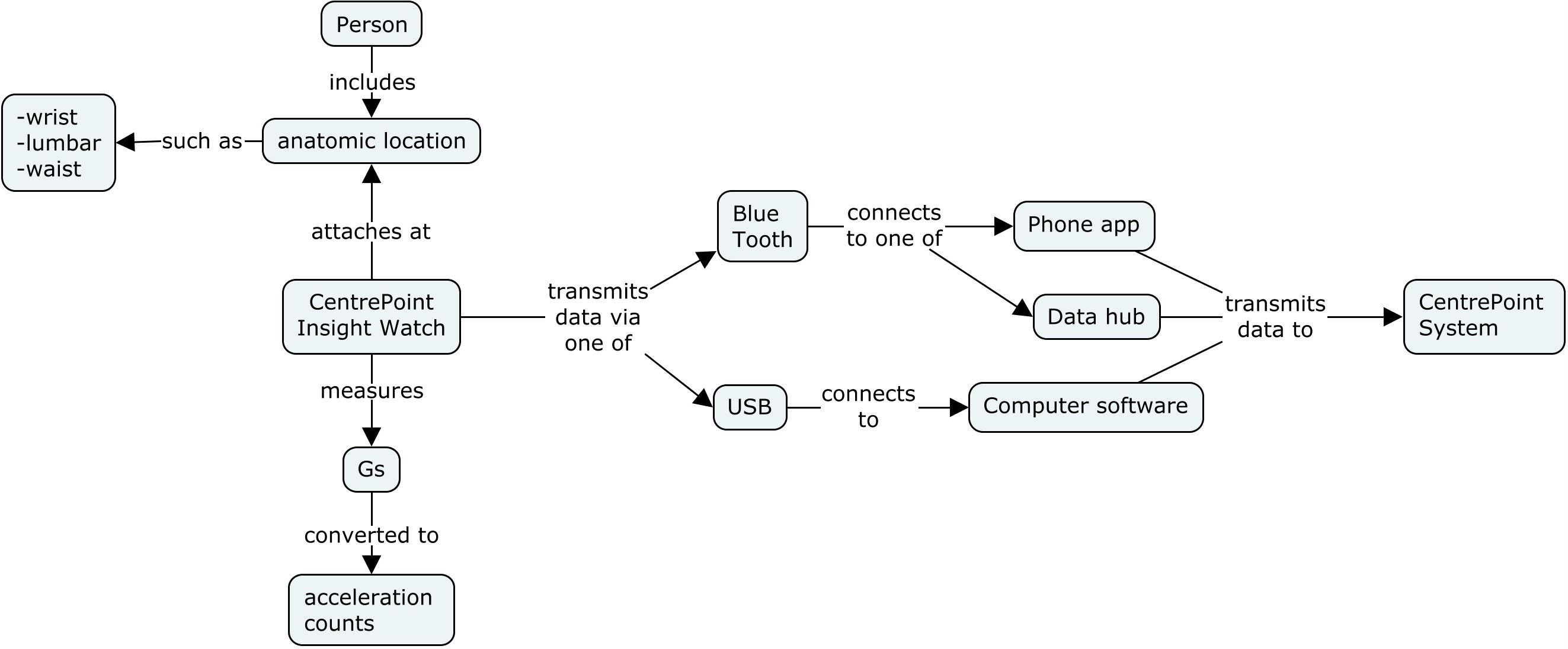
This concept map shows the components of the actigraphy device, as well as various devices that can be used to transmit data to the Centrepoint System, where data are processed.
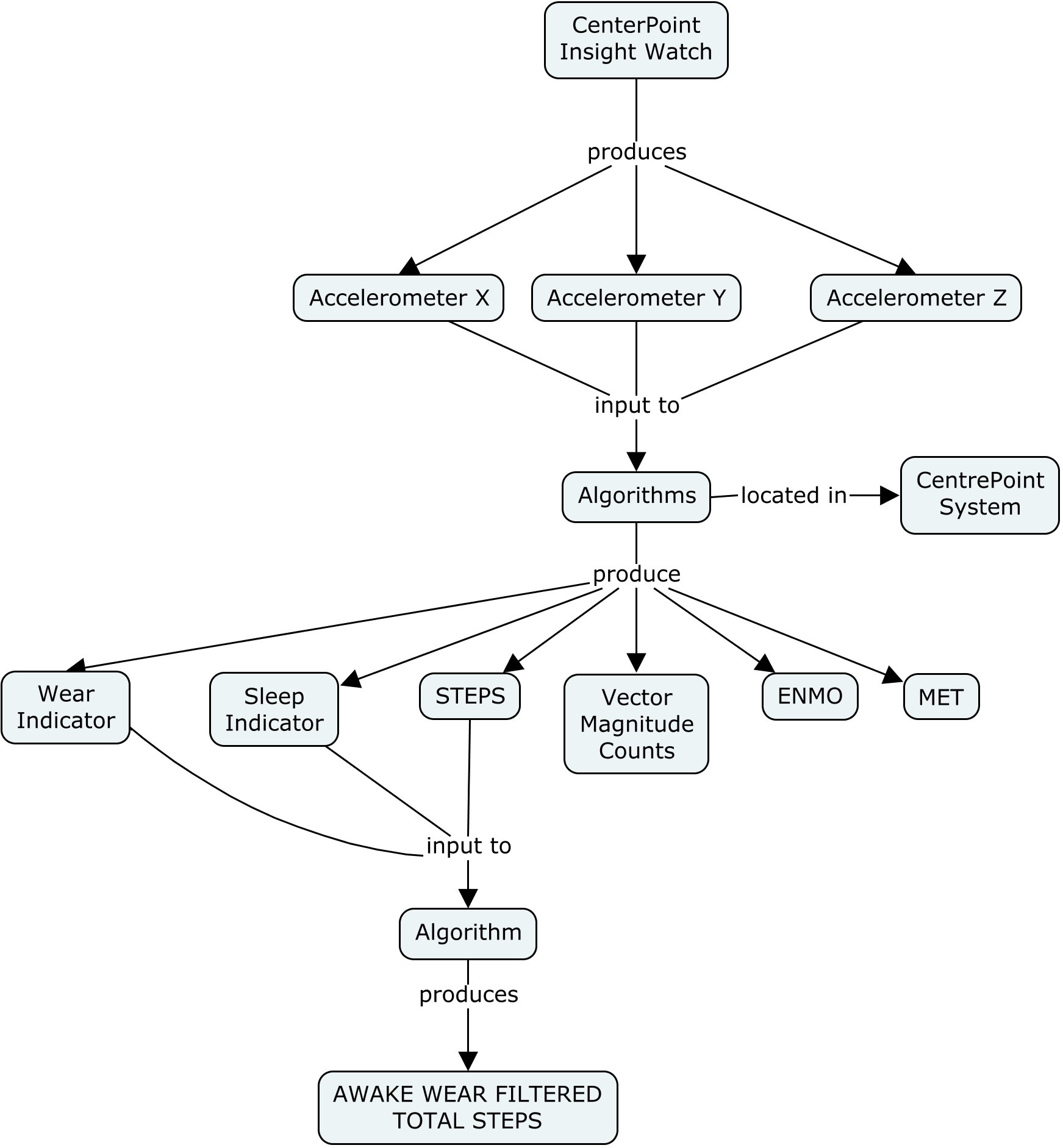
This additional concept map shows how the data are processed.
This concept map shows the actigraphy devices and the step count data produced as biomedical concepts.
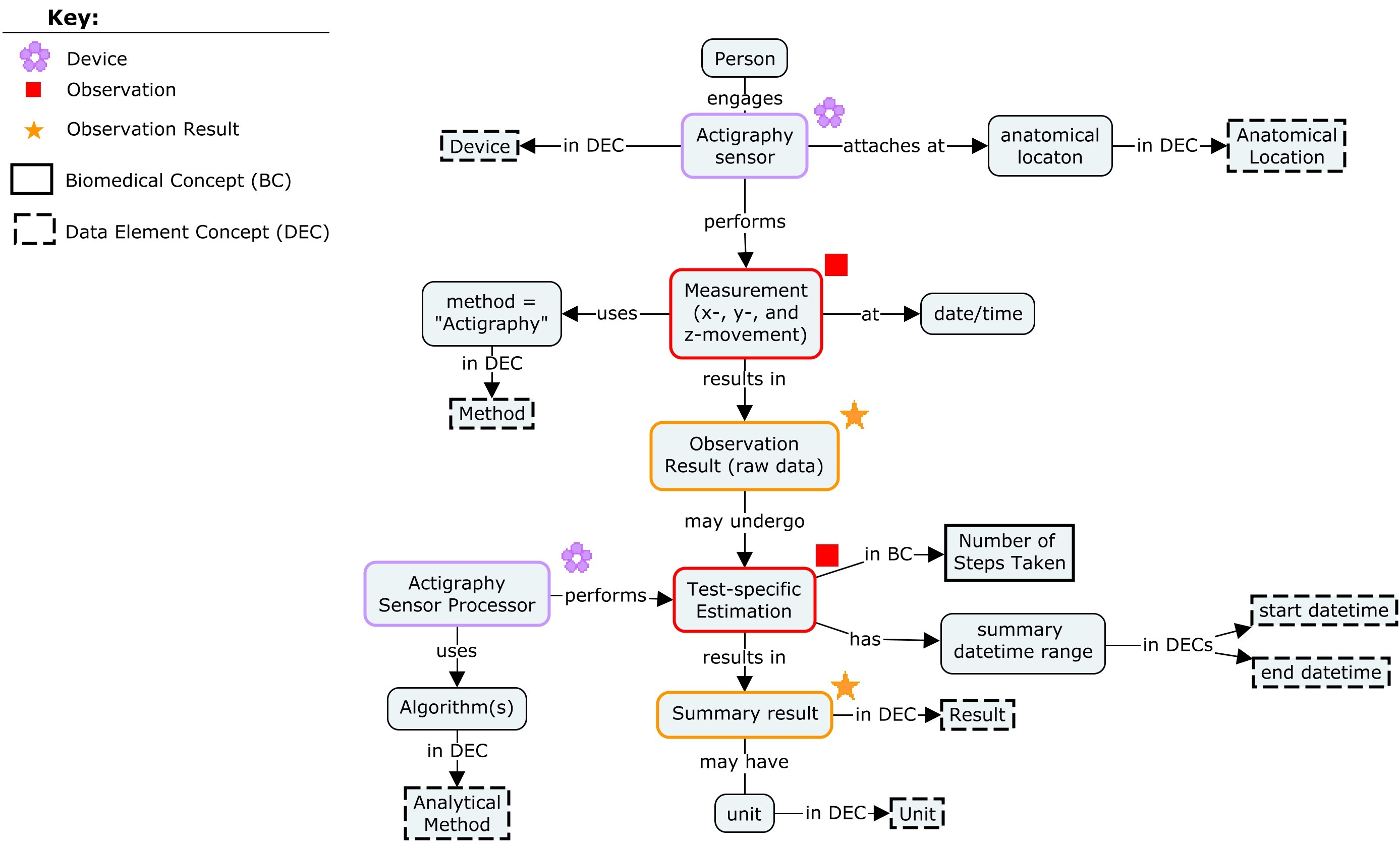
The devices are considered separate biomedical concepts. They are represented in the SDTM datasets Device Identifiers (DI) and Related Devices (RELDEV).
The step count data produced by the device is a biomedical concept which is represented in an SDTM Findings domain.
The name of the findings biomedical concept maps to the SDTM variables --TESTCD and --TEST. The device in this example captured total steps during and day and also monitored whether the device was being worn and whether the subject was asleep. The measurement provided by the device was "awake wear filtered total steps."
This table shows the mapping of the data element concepts (DECs) to the variables in the SDTM Findings domain.
| Device | SPDEVID |
| Method | MKMETHOD |
| Analytical Method | MKANMETH |
| Result | MKORRES, MKSTRESC, MKSTRESN |
| Unit | MKORRESU, MKSTRESU |
| Start datetime summary range | MKDTC |
| End datetime summary range | MKENDTC |
The following should be considered when viewing this resource:
- The Device Identification Parameter "Device Type" is defined as "Generic category of medical device within the device classification established by a regulatory authority." This suggests that terms should be taken from a vocabulary established by a regulatory authority. It is not clear what vocabulary should be used.
- Step count data was represented in the Musculoskeletal System Findings (MK) domain. Ambulation is not the product of the musculoskeletal system alone, so this choice of domain could be changed in future.
- The TESTCD and TEST values "STEPSTN" and "Number of Steps Taken" from the Findings About Test Code and Findings About Test Name codelists were used to represent the number of steps, although It is not clear that the meaning of the "awake wear filtered steps" fits the definition of this test, which is, "The number of steps taken by an individual." The "Number of Steps Taken" test name and code are not in the Musculoskeletal Findings Test Name and Musculoskeletal Findings Test Code codelists.
- The root variable --REFID is defined as, "A sequence of characters used to uniquely identify a source of information." In this example, MKREFID is the "DEVICEID" from the dataset provided by the device vendor. An alternative approach would have been to define a separate identifier (SPDEVID) for each individual device.
- In this example, three specific algorithms involved in the processing of the raw data were identified and were used to populate MKANMETH. SDTM terminology includes "Analytical Method Calculation Formula" codelists associated with the --ANMETH variable for certain other SDTM domains, but there is not currently a codelist associated with the MKANMETH domain.
- Algorithms can also be modeled as properties of the device, but that would not allow the identification of the different algorithms used for a specific test.
- The root variable --LOC is defined as, "The anatomical location of the intervention, event, or assessment." MKLOC was used to represent the location at which the device was worn, under the assumption that the location at which the device was worn is the location of the assessment.
- The study referenced by DiMe Endpoint 399 used a "hip-worn" device, but the sample data used to construct this example was collected by a wrist-worn watch, so MKLOC was populated with "WRIST." The term "WRIST JOINT" is in the SDTM Anatomical Location (LOC) codelist, but "WRIST" is not.
- Note that although there is a unit "STEPS" in the SDTM UNIT codelist, this is a measure of distance, as in "The tree is 12 steps from my front door." The SDTM test "Number of Steps Taken" includes what is being counted in the name of the test, so in a record for this test the SDTM variables --ORRESU and --STRESU are null. * Some terminology used in the biomedical concepts is present in the NCI Thesaurus, as reflected in C-codes included in biomedical concepts and SDTM specializations. If an appropriate C-code is not available, the placeholder "Cnew" is used.
- Some SDTM variable values used in the example SDTM datasets are included in CDISC SDTM terminology, but in other cases where a codelist is associated with an SDTM variable, the values used in the example are not in the codelist.
Known Issue
The measurement provided by the device, "awake wear filtered total steps" is a count of steps during the time period which the device determines that the device is being worn and the subject is awake. It is clear that the amount of time the device was worn over the course of a day affects how the number of steps recorded while the device was worn. It is less obvious how the subject being awake or asleep affects the number of "awake aware filtered steps." In this example, it seemed necessary to provide "awake aware minutes" to assist in understanding "awake aware filtered steps". The approach used in this example was to use two MK domain tests, annot be usefully interpreted unless one knows was represented as two tests in the MK domain, "Number of Steps" with an evaluation interval of "AWAKE WEAR MINUTES IN DAY" and "Awake Wear Minutes" with an evaluation interval of "CALENDAR DAY". This solution is less than satisfying, as the awake wear minutes are primarily a measure of how much the device was worn, which is not a finding about the subject's musculoskeletal system. It is more a measure of the device's operation, but is hard to represent in the current Device Exposure (DE) domain, which is designed to represent intervals of time in which the subject is exposed to the device, and this is a summary of the durations of periods of non-exposure. Because of the questions about whether "Awake Wear Minutes" is an appropriate Musculoskeletal Finding the test "Awake Wear Minutes" was not developed as a biomedical concept.
This approach chosen in this example is similar, in some ways, to the use of the test, "Technical Quality" in the ECG and Holter ECG test name codelists.
The example DI dataset shows how the devices modelled are described (parameters used) and the device identifiers (SPDEVID values) given to the devices.
The sponsor in this study chose to use a "kind of" device identifier for the CentrePoint Insight Watch rather than an identifier for each individual watch.
For a software system, version can be an identifying characteristic. However, Ametris uses the same software version throughout any study, so it is not needed in this example.
Since there is only identifier for each of the component devices, there is only one identifier for the combined device.
di.xpt
Rows 1-3: Show characteristics of the CentrePoint Insight Watch as parameters and assign it the identifer (SPDEVID) "CPIW".
Rows 4-6: Provide the characteristics of the CentrePoint System and assign it the SPDEVID "CentrePoint System". The value used for the SPDEVID is the sponsor's choice. It can the device's full name, as in this case, or an abbreviation, or an arbitrary string of characters.
Rows 7-0: Provide the characteristics of the combined device and assign it the SPDEVID "CentrePoint".
| Row | STUDYID | DOMAIN | SPDEVID | DISEQ | DIPARMCD | DIPARM | DIVAL |
|---|---|---|---|---|---|---|---|
| 1 | 757 | DI | CPIW | 1 | DEVTYPE | Device Type | Actigraphy Sensor |
| 2 | 757 | DI | CPIW | 2 | MANUF | Manufacturer | Ametris L.L.C. |
| 3 | 757 | DI | CPIW | 3 | TRADENAM | Trade Name | CentrePoint Insight Watch |
| 4 | 757 | DI | CentrePoint System | 1 | DEVTYPE | Device Type | Actimetry Signal Processor |
| 5 | 757 | DI | CentrePoint System | 2 | MANUF | Manufacturer | Ametris L.L.C. |
| 6 | 757 | DI | CentrePoint System | 3 | TRADENAM | Trade Name | CentrePoint System |
| 7 | 757 | DI | CentrePoint | 1 | DEVTYPE | Device Type | Physical Activity Monitoring System |
| 8 | 757 | DI | CentrePoint | 2 | MANUF | Manufacturer | Ametris L.L.C. |
| 9 | 757 | DI | CentrePoint | 3 | TRADENAM | Trade Name | CentrePoint |
Relevant step count data from the device output file have been mapped to the following Musculoskeletal System Findings (MK) domain dataset.
- SPDEVID (Sponsor Device Identifier) is defined in the Device Identifier (DI) above. In this study, the implementer chose to use an identifier that identified the kind of device, rather than the individual device.
- LBREFID is populated with an identifier for the individual device supplied by the device vendor.
- The step count result provided by the device vendor can be fully described as "the total number of steps takenby the subject during a one-day period during times when the subject was awake and the device was worn." In the SDTM record, this complete description is represented by a combination of variables:
- MKTEST and MKTESTCD represent "number of steps taken"
- MKCOLSRT represents "total"
- MKSTDTC and MKENDTC represent the start and end of the one-day monitoring period
- MKEVINTX represents the time periods considered in calculation of the result, the awake wear minutes in the day.
- The amount of time summarized by the step count is represented by
- MKTEST and MKTESTCD represent the time counted, "AWAKE WEAR" minutes.
- MKCOLSRT represents "total."
- MKSTDTC and MKENDTC represent the start and end of the one-day monitoring period.
- MKEVINTX represents the time period considered in calculation of the result, "CALENDAR DAY".
mk.xpt
Rows 1-6: Show numbers of awake-wear filtered steps and number of awake-wear minutes for subject 81999 on three successive days.
Rows 7-12: Show numbers of awake-wear filtered steps and number of awake-wear minutes for subject 90507 on three different days.
Rows 13-14: Show a day for subject 90507 with 90 minutes when the subject was awake and wearing the device, but took zero steps during that time.
| Row | STUDYID | DOMAIN | USUBJID | SPDEVID | MKSEQ | MKGRPID | MKREFID | MKTESTCD | MKTEST | MKORRES | MKCOLSRT | MKLOC | MKMETHOD | MKANMETH | MKDTC | MKEVINTX | MKANMTH1 | MKANMTH2 | MKANMTH3 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 757 | MK | 81999 | CentrePoint | 1 | 1 | CPW1B22200290 | STEPSTN | Number of Steps Taken | 10618 | TOTAL | WRIST | ACTIGRAPHY | MULTIPLE | 2023-03-01 | AWAKE WEAR MINUTES IN DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM | DELGADO-GONZALO 2015 STEP COUNT ESTIMATE ALGORITHM | |
| 2 | 757 | MK | 81999 | CentrePoint | 2 | 1 | CPW1B22200290 | AWKWEAR | Awake Wear Minutes | 795 | TOTAL | WRIST | ACTIGRAPHY | MULTIPLE | 2023-03-01 | CALENDAR DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM | ||
| 3 | 757 | MK | 81999 | CentrePoint | 3 | 2 | CPW1B22200290 | STEPSTN | Number of Steps Taken | 5006 | TOTAL | WRIST | ACTIGRAPHY | MULTIPLE | 2023-03-02 | AWAKE WEAR MINUTES IN DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM | DELGADO-GONZALO 2015 STEP COUNT ESTIMATE ALGORITHM | |
| 4 | 757 | MK | 81999 | CentrePoint | 4 | 2 | CPW1B22200290 | AWKWEAR | Awake Wear Minutes | 495 | TOTAL | WRIST | ACTIGRAPHY | MULTIPLE | 2023-03-02 | CALENDAR DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM | ||
| 5 | 757 | MK | 81999 | CentrePoint | 5 | 3 | CPW1B22200290 | STEPSTN | Number of Steps Taken | 2423 | TOTAL | WRIST | ACTIGRAPHY | MULTIPLE | 2023-03-03 | AWAKE WEAR MINUTES IN DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM | DELGADO-GONZALO 2015 STEP COUNT ESTIMATE ALGORITHM | |
| 6 | 757 | MK | 81999 | CentrePoint | 6 | 3 | CPW1B22200290 | AWKWEAR | Awake Wear Minutes | 840 | TOTAL | WRIST | ACTIGRAPHY | MULTIPLE | 2023-03-03 | CALENDAR DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM | ||
| 7 | 757 | MK | 90507 | CentrePoint | 3 | 2 | CPW1B34200297 | STEPSTN | Number of Steps Taken | 127 | TOTAL | WRIST | ACTIGRAPHY | MULTIPLE | 2023-06-21 | AWAKE WEAR MINUTES IN DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM | DELGADO-GONZALO 2015 STEP COUNT ESTIMATE ALGORITHM | |
| 8 | 757 | MK | 90507 | CentrePoint | 4 | 2 | CPW1B34200297 | AWKWEAR | Awake Wear Minutes | 502 | TOTAL | WRIST | ACTIGRAPHY | MULTIPLE | 2023-06-21 | CALENDAR DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM | ||
| 9 | 757 | MK | 90507 | CentrePoint | 5 | 3 | CPW1B34200297 | STEPSTN | Number of Steps Taken | 2114 | TOTAL | WRIST | ACTIGRAPHY | MULTIPLE | 2023-06-24 | AWAKE WEAR MINUTES IN DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM | DELGADO-GONZALO 2015 STEP COUNT ESTIMATE ALGORITHM | |
| 10 | 757 | MK | 90507 | CentrePoint | 6 | 3 | CPW1B34200297 | AWKWEAR | Awake Wear Minutes | 643 | TOTAL | WRIST | ACTIGRAPHY | MULTIPLE | 2023-06-24 | CALENDAR DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM | ||
| 11 | 757 | MK | 90507 | CentrePoint | 7 | 4 | CPW1B34200297 | STEPSTN | Number of Steps Taken | 405 | TOTAL | WRIST | ACTIGRAPHY | MULTIPLE | 2023-06-25 | AWAKE AWARE MINUTES IN DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM | DELGADO-GONZALO 2015 STEP COUNT ESTIMATE ALGORITHM | |
| 12 | 757 | MK | 90507 | CentrePoint | 8 | 4 | CPW1B34200297 | AWKWEAR | Awake Wear Minutes | 494 | TOTAL | WRIST | ACTIGRAPHY | ALGORITHM Y | 2023-06-25 | CALENDAR DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM | ||
| 13 | 757 | MK | 90507 | CentrePoint | 15 | 8 | CPW1B34200297 | STEPSTN | Number of Steps Taken | 0 | TOTAL | WRIST | ACTIGRAPHY | ALGORITHM X | 2023-09-01 | AWAKE WEAR MINUTES IN DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM | DELGADO-GONZALO 2015 STEP COUNT ESTIMATE ALGORITHM | |
| 14 | 757 | MK | 90507 | CentrePoint | 16 | 8 | CPW1B34200297 | AWKWEAR | Awake Wear Minutes | 90 | TOTAL | WRIST | ACTIGRAPHY | ALGORITHM Y | 2023-09-01 | CALENDAR DAY | CHOI 2011 WEAR AND NONWEAR ALGORITHM | VAN HEES 2018 SLEEP INTERRUPTION ALGORITHM |
| Variable | Label | Type | Role |
|---|---|---|---|
| MKANMTH1 | Analysis Method 1 | Char | Non-Standard Qualifier |
| MKANMTH2 | Analysis Method 2 | Char | Non-Standard Qualifier |
| MKANMTH3 | Analysis Method 3 | Char | Non-Standard Qualifier |
References
Accelerometry Data, General:
Accelerometry data in health research: challenges and opportunities - PMC Stat Biosci. 2019 Jan 12;11(2):210–237.
Steps:
R. Delgado-Gonzalo et al., “Physical Activity Profiling: Activity-Specific Step Counting and Energy Expenditure Models Using 3D Wrist Acceleration,” in 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) (2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan: IEEE, 2015), 8091–94, https://doi.org/10/gpn5sj.
Wear/NonWear:
Leena Choi et al., “Validation of Accelerometer Wear and Nonwear Time Classification Algorithm,” Medicine & Science in Sports & Exercise 43, no. 2 (February 2011): 357–64, https://doi.org/10/bzww86.
Sleep:
Vincent Theodoor van Hees et al., “Estimating Sleep Parameters Using an Accelerometer without Sleep Diary,” Scientific Reports 8, no. 1 (August 28, 2018): 12975, https://doi.org/10.1038/s41598-018-31266-z.
DHT Portal Steps Example.xlsx