Introduction
The CDISC Analysis Data Model Implementation Guide (ADaMIG) provides several timing variables for modeling clinical trial designs in analysis datasets. APHASE, APERIOD, and ASPER can be used in conjunction with related treatment variables to meet a variety of analysis requirements, from single-period parallel studies to much more complicated situations involving multiple treatment periods and even different studies. The goal of this article is to provide guidelines for identifying when to use the different timing variables that are available. Additional examples can be found in the References and Resources listed at the end of the article.
Advice, Instructions, and Guidance
The main reason for the creation of the primary timing variables in ADaM is the need for analyzing study results using different time divisions than what has been collected or modeled in SDTM using EPOCH. For example, the SAP may define visit or time-division windowing in order to guarantee that observations collected at a similar time interval are summarized together, regardless of the CRF page on which they were recorded. Additionally, analysis time divisions may view the study epochs differently than how the data were collected. ADaM timing variables should reflect the analysis requirements, and not simply copy values from SDTM epochs or visits.
The ADaMIG provides the following standard variables, along with their corresponding numeric or character counterparts, which can be used in ADaM datasets to represent different types of epochs or time divisions within a study:
APHASE (Phase) represents the analysis phase associated with the record, which is a study time division that may or may not involve the administration of an investigational product. Examples of APHASE values are Screening, Treatment and Follow-up, but there is no controlled terminology associated with APHASE, so anything that reflects the wording displayed on the tables is acceptable.
APERIOD (Period) represents the analysis period associated with the record. As noted in the ADaMIG, the value of APERIOD must correspond to one of the analysis periods defined by the TRTxxP variables in ADSL. APERIOD contains a numeric value ranging from 1 to 99.
ASPER (Subperiod within Period) denotes a subperiod within an analysis period. It also contains a numeric value; the first subperiod within a period is always assigned a value of 1, and the counter starts again for any subperiods defined within subsequent analysis periods.
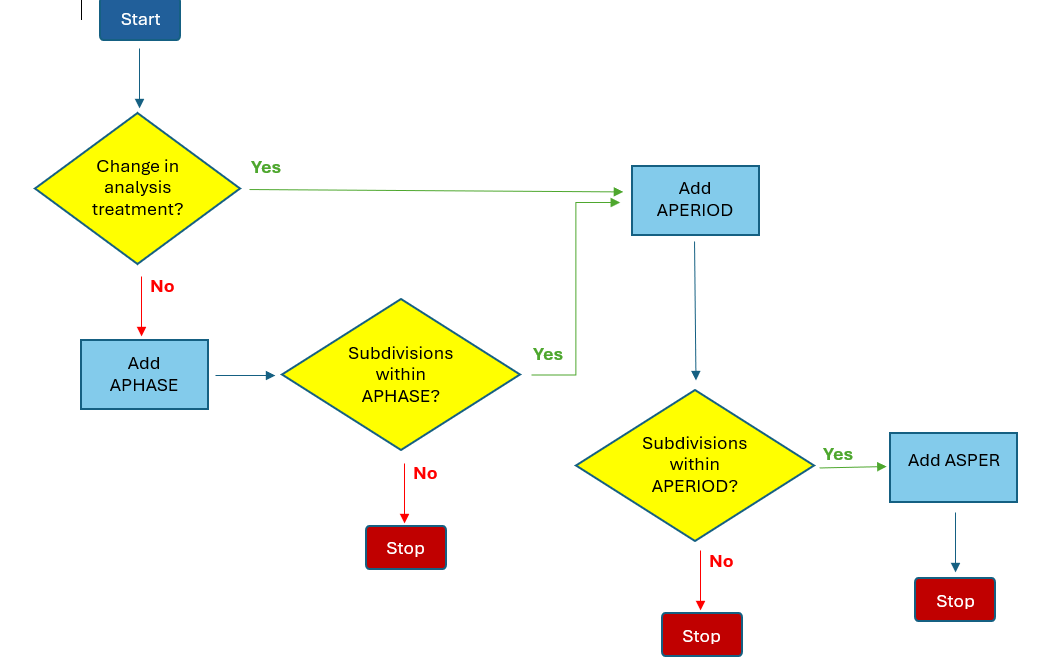
The first step in deciding which of these variables to use is to determine whether there is a change in analysis study treatment when a subject moves between study epochs. If there is no study treatment involved, or the treatment assignment does not change when the next study epoch begins, then APHASE should generally be used to represent the study division needed for analysis. If the subject moves from one treatment to another when the study division changes, then APERIOD should generally be used, especially if the treatment is represented by one of the TRTxxP variables in ADSL. Finally, if the study division represents a subset of an analysis period, then ASPER is generally appropriate.
Examples
The following flowchart models these decision rules.
Suppose you have a crossover study with 2 treatment periods:
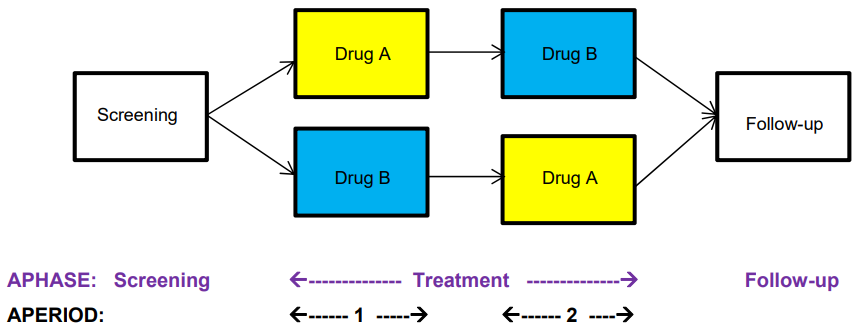
Since the study treatment changes during each of the crossover epochs, this design can be modeled by assigning each of the treatment epochs to a different analysis period (APERIOD). Note that APERIOD is numeric. If desired, we could also create APERIODC to contain a character description of APERIOD; there must be a 1:1 relationship between the values of APERIOD and APERIODC within a dataset when both are populated. However, APERIOD is considered to be the primary variable, so APERIODC may only be used when APERIOD is also present. Note that. again, we only need to create APHASE when we need to summarize screening, treatment, and follow-up records separately. If we are only summarizing treatment records, then simply using APERIOD will suffice.
Additional examples, including study designs with washout periods, multiple progressions, subperiods and treatment cycles can be found in https://www.lexjansen.com/pharmasug/2019/DS/PharmaSUG-2019-DS-250.pdf
To summarize, ADaMIG defines the variables APERIOD, ASPER, and APHASE, which can be used for modeling different types of study designs in both ADaM Basic Data Structure (BDS) and Occurrences (OCCDS) datasets, based on analysis requirements. The flowchart may be helpful in determining which variables to use for a given study, depending on the design and analysis needs. The first question to answer is whether there is a change in analysis treatment from one study segment to the next. The response to that question determines where to proceed from there, and should help guide you to the correct choice of variables to use in modeling your study design.
References
ADaMIG v1.3, Section 3.3.3, Timing Variables for BDS Datasets
ADaMIG v1.3, Section 4.8, Examples of Timing Variables
Resources
Brucken N. Timing is everything: defining ADaM period, subperiod and phase (Paper DS-250). PharmaSUG;2019. Accessed March 9, 2026. https://www.lexjansen.com/pharmasug/2019/DS/PharmaSUG-2019-DS-250.pdf
Fulton J. CDISC ADaM phases, periods, and subperiods: a case study (Paper DS-014). PharmaSUG;2021. Accessed March 9, 2026. https://pharmasug.org/proceedings/2021/DS/PharmaSUG-2021-DS-014.pdf
Zhu S. Implementation of ADaM Basic Data Structure for cross-over studies (Paper DS13). PharmaSUG;2016. Accessed March 9, 2026. https://pharmasug.org/proceedings/2016/DS/PharmaSUG-2016-DS13.pdf