The visualization below describes key digital health technologies (DHT) concepts and their relationships in the context of the clinical research information lifecycle. Controlled terminology is currently available for a subset of concepts and is:
- Published in the DiMe Glossary
- Under consideration for the CDISC Glossary
Description
An individual who may play a specific role in a clinical research setting engages with a DHT. This engagement has attributes such as whether an individual's interaction with the device was active or passive, and when and where the engagement occurred. The DHT in use also has attributes such as its status in relation to regulators and mechanisms for data capture. In the context of clinical research, the measurements collected may be used for many purposes including as biomarkers and for the generation of outcome assessments.
Key DHT Concepts
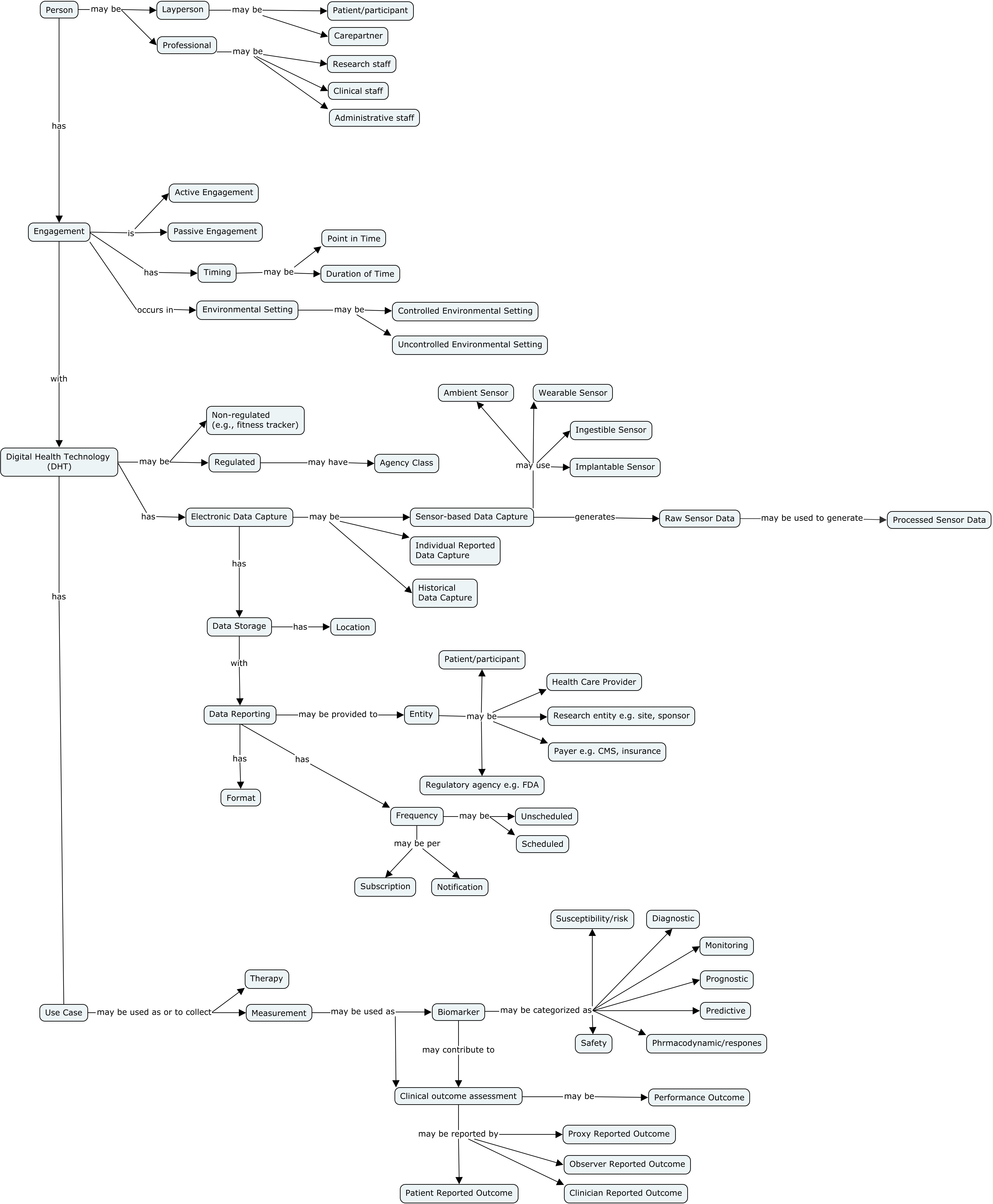
Table 1. DRAFT DHT Glossary developed by the DHT team. These terms and definitions will be integrated into the CDISC Glossary in future.
| NCI C-code | CDISC Preferred Term | CDISC Synonym | CDISC Definition |
|---|---|---|---|
| CNEW | Digital Health Technology | Electronic tools, systems, devices and resources that generate, store and process data in health care and research, which may include mobile health, wearable devices, telehealth, telemedicine, electronic health records, and patient portals. (DiMe) | |
| C142527 | Electronic Data Capture | The process of collecting health data into an electronic form. | |
| CNEW | Sensor-Based Data Capture | A process designed to collect data from a transducer that converts a physical, biological, or chemical parameter into an electrical signal, which is then converted into an electronic form. | |
| CNEW | Individual Reported Data Capture | A process designed to collect data in an electronic form directly from an individual, such as a patient, caregiver, clinician, or observer, by a direct interaction with a tool. | |
| CNEW | Historical Data Capture | A process designed to collect data in an electronic form from previously collected data. | |
| CNEW | Ambient Sensor | A transducer that converts a physical, biological, or chemical parameter into an electrical signal, which functions outside of the body. | |
| CNEW | Wearable Sensor | A transducer that converts a physical, biological, or chemical parameter into an electrical signal, which functions in contact with the body. | |
| CNEW | Ingestible Sensor | A transducer that converts a physical, biological, or chemical parameter into an electrical signal, which functions as it travels through the gastrointestinal tract. | |
| CNEW | Implantable Sensor | A transducer that converts a physical, biological, or chemical parameter into an electrical signal, which functions when integrated with tissue in the body. | |
| CNEW | Raw Sensor Data | Pre-processed Data | Data generated from a digital sensor that may have been through some hardware and/or software filtering but are generally considered to be unprocessed. |
| CNEW | Processed Sensor Data | Data generated from raw sensor data that has been refined and structured through additional operations such as data cleaning, compression, and algorithmic transformation to generate clinically interpretable data. | |
| C142378 | Clinical Outcome Assessment (COA) | Any assessment that may be influenced by human choices, judgment, or motivation and may support or refute treatment benefit. NOTE: Unlike biomarkers that rely completely on an automated process or algorithm, COAs reflect interpretation of reporting from a patient, a clinician, or an observer. A COA may be based on reports by persons or on performance of tasks. (CDISC Glossary) | |
| C142632 | Performance Outcome (PerfO) | A measurement based on standardized task(s) performed by a patient that is administered and evaluated by an appropriately trained individual or is independently completed. (BEST (Biomarkers, Endpoints and Other Tools) Resource) | |
| C95401 | Patient-reported Outcome (PRO) | A measurement based on a report that comes directly from the patient about the status of a patient's health condition without amendment or interpretation of the patient's response by a clinician or anyone else. (BEST (Biomarkers, Endpoints and Other Tools) Resource) | |
| C142453 | Clinician-reported Outcome (ClinRO) | A measurement based on a report that comes from a trained health-care professional after observation of a patient's health condition. (BEST (Biomarkers, Endpoints and Other Tools) Resource) | |
| C142620 | Observer-reported Outcome (ObsRO) | A measurement based on a report of observable signs, events or behaviors related to a patient's health condition by someone other than the patient or a health professional. (BEST (Biomarkers, Endpoints and Other Tools) Resource) | |
| C142653 | Proxy-Reported Outcome | A measurement based on a report by someone other than the patient reporting as if he or she is the patient. (CDISC Glossary) | |
| CNEW | Active Engagement | A purposeful interaction in which the individual is engaged in the activity with intentional involvement. | |
| CNEW | Passive Engagement | A purposeful interaction in which the individual is engaged in the activity without intentional involvement. | |
| C188840 | Environmental Setting | The environment or physical setting where the event, intervention, or finding occurred. | |
| CNEW | Controlled Environmental Setting | An environmental setting in which specific conditions are maintained within a purpose-built space. | |
| CNEW | Uncontrolled Environmental Setting | An environmental setting that is not purpose-built, managed, or maintained by an entity. |