This example shows findings from assessments of from a continuous glucose monitor (CGM) with the purpose of supporting DIME Endpoint 125, "CGM % Time 70-180 mg/dl," in the regulatory submission of a clinical trial. The CGM data of interest are defined in the study protocol.
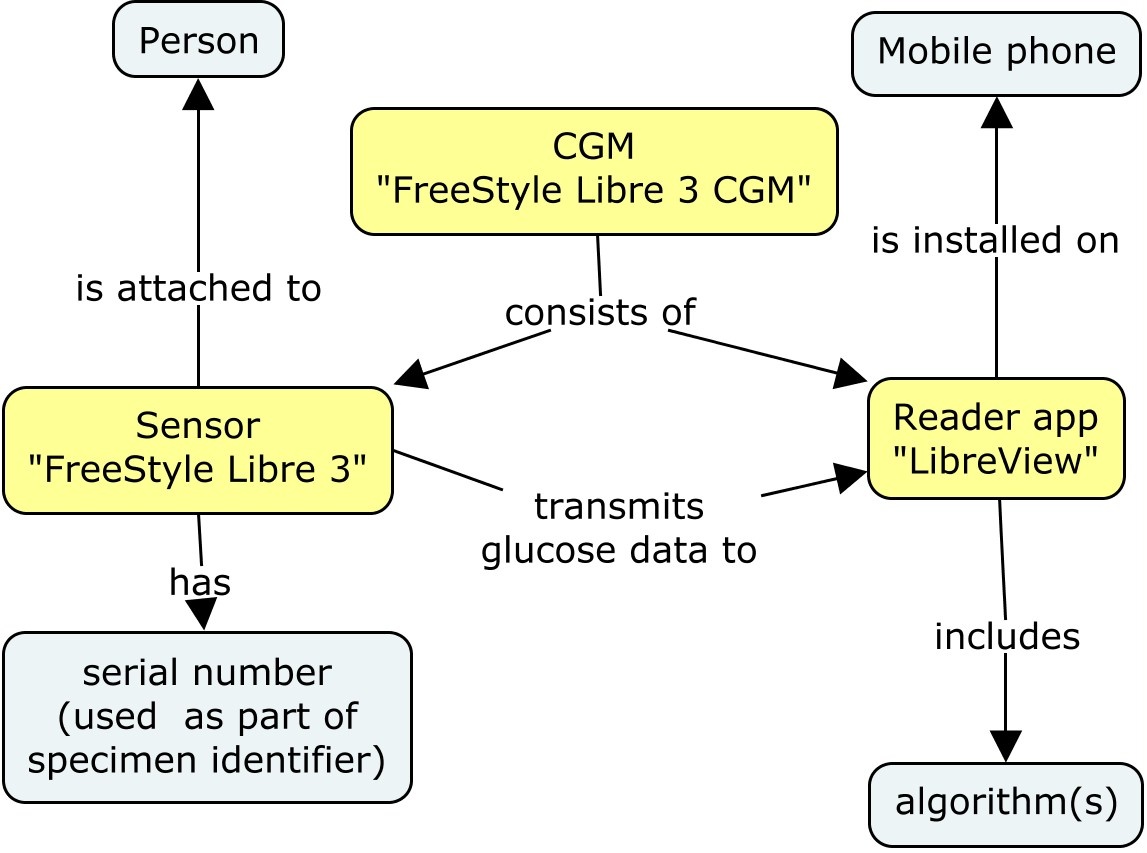
This concept map shows the modeling of the CGM device and its components. Devices shown in yellow are those represented in the DI and RELDEV domains. The concept map includes other entities ("bubbles") to aid in the understanding of how the device works. For this study, it was not necessary to represent the further device components shown in the concept map in the device datasets.
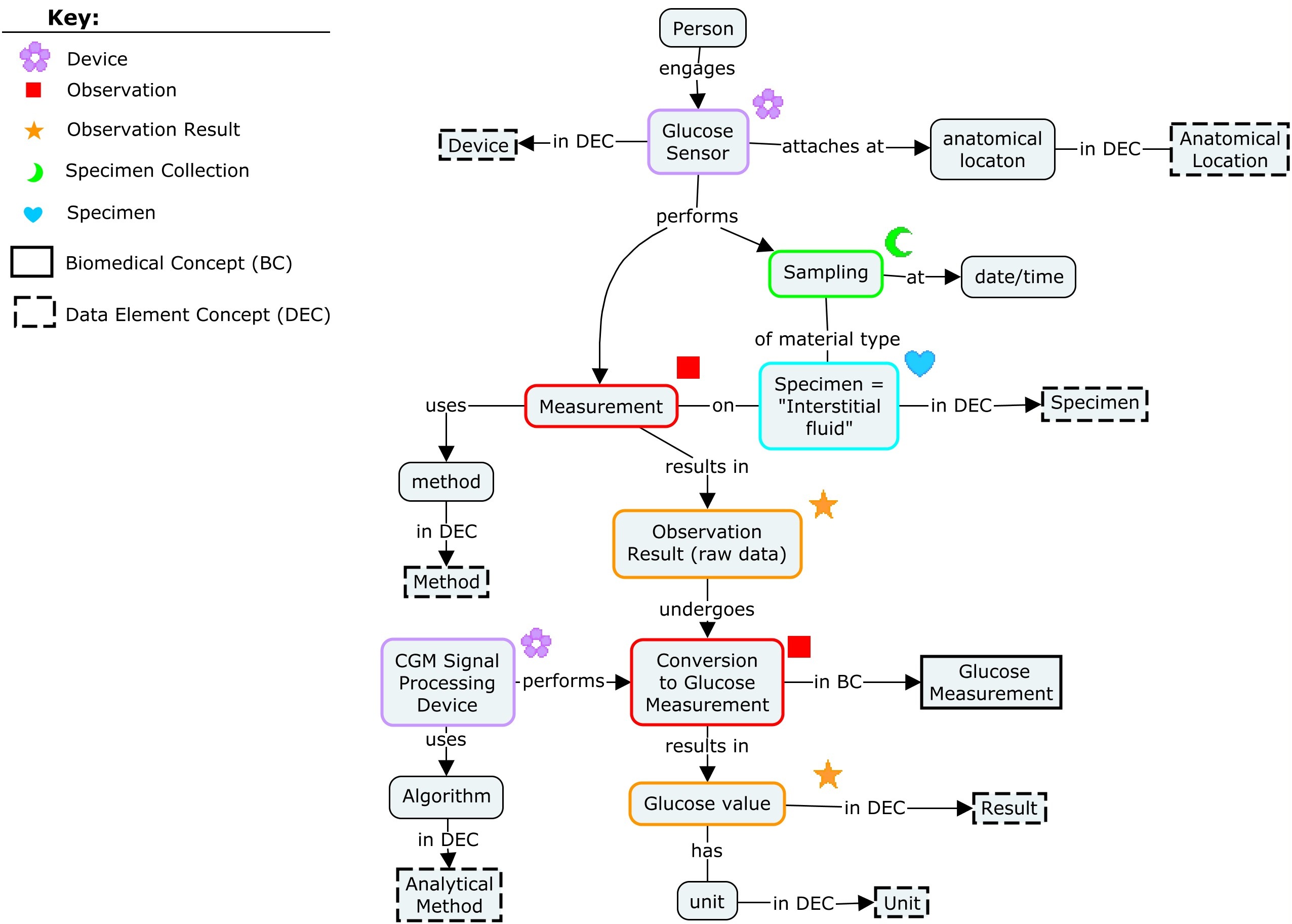
This concept map shows the CGM devices and the glucose data produced by the CGM as biomedical concepts.
The CGM devices are considered separate biomedical concepts. They are represented in the SDTM datasets Device Identifiers (DI) and Related Devices (RELDEV).
The measurement produced by the device is a biomedical concept which is represented in an SDTM Findings domain.
The name of the Biomedical Concept maps to the SDTM variables --TESTCD and --TEST. For this CGM, the BC name "Glucose Measurement" maps to the LBTESTCD "GLUCPE" and LBTEST "Plasma Equivalent Glucose"
This table shows the mapping from the data element concepts (DECs) that comprise the biomedical concepts to the variables in the SDTM Findings domain.
| Device | SPDEVID |
| Specimen | LBSPEC |
| Method | LBMETHOD |
| Analytical Method | LBANMETH |
| Result | LBORRES, LBSTRESC, LBSTRESN |
| Unit | LBORRESU, LBSTRESU |
The following should be considered when viewing this resource:
- The definition of the Laboratory Test Results (LB) domain is currently, "A findings domain that contains laboratory test data such as hematology, clinical chemistry and urinalysis. This domain does not include microbiology or pharmacokinetic data, which are stored in separate domains." This example assumes that "laboratory test data" includes tests performed outside of a laboratory, and ones in which the specimen tested is not removed from the body.
- The Device Identification Parameter "Device Type" is defined as "Generic category of medical device within the device classification established by a regulatory authority." This suggests that terms should be taken from a vocabulary established by a regulatory authority. It is not clear what vocabulary should be used.
- The variable LBLOC was used for the location at which the sensor was attached. The definition of the root variable --LOC is, "The anatomical location of the intervention, event, or assessment." It seems reasonable to model the location of the assessment as the location at which the sensor is attached.
- The variable LBANMETH was not included in this example. It is not clear when it would be included in an LB dataset and, if it is included, what its value should be.
- The device output file includes "Serial Number." Since sensors are replaced every 14 days, we assumed that this is a serial number for the sensor and that it is appropriate to use in forming LBREFID. The definition of the root variable --REFID is, "A sequence of characters used to uniquely identify a source of information."
- The example LB dataset above does not include variables for normal ranges, but normal range variables would likely be included in a regulatory submission. For a validated device, the normal ranges would be supplied by the device manufacturer. and specified in the protocol. For non-validated devices, there may not be a known normal range.
The example DI dataset shows how the devices modelled are described (parameters used) and the device identifiers (SPDEVID values) given to the devices. Note that the parameters for a device describe it to the level of granularity needed for the particular trial.
di.xpt
Rows 1-2: Describe the device given the sponsor identifier "Libre View"
Rows 3-4: Describe the device given the sponsor identifier "FreeStyle Libre 3". Note that although the serial number of this device is used in the specimen identifier (LBREFID) in the LB domain, identifying individual devices is not necessary in the device data for this study.
Rows 5-6: Describe the device given the sponsor identifier "FreeStyle Libre 3 CGM".
di.xpt
| Row | STUDYID | DOMAIN | SPDEVID | DISEQ | DIPARMCD | DIPARM | DIVAL |
|---|---|---|---|---|---|---|---|
| 1 | ABC | DI | Libre View | 1 | DEVTYPE | Device Type | Reader app |
| 2 | ABC | DI | Libre View | 1 | MANUF | Manufacturer | Abbot |
| 3 | ABC | DI | FreeStyle Libre 3 | 1 | DEVTYPE | Device Type | Sensor |
| 4 | ABC | DI | FreeStyle Libre 3 | 1 | MANUF | Manufacturer | Abbot |
| 5 | ABC | DI | FreeStyle Libre 3 CGM | 1 | DEVTYPE | Device Type | CGM System |
| 6 | ABC | DI | FreeStyle Libre 3 CGM | 1 | MANUF | Manufacturer | Abbot |
Glucose data were mapped to the SDTMIG Laboratory Test Results (LB) domain. This example includes the four glucose values in the device output file; the record type (historic or scan) does not matter for the endpoint and is not captured in the SDMT LB dataset.
- The Sponsor Device Identifier (SPDEVID) is assigned in the Device Identifiers (DI) dataset above.
- LBREFID is a sample identifier, created by concatenating the device serial number and the datetime. The variable --REFID is defined as, "A sequence of characters used to uniquely identify a source of information" and is used to establish traceability of the results to a source. In this case, the source is a particular row in the device output file.
- LBDTC is the device timestamp represented in the ISO 8601 format used by SDTM.
lb.xpt
lb.xpt
| Row | STUDYID | USUBJID | SPDEVID | LBSEQ | LBREFID | LBTESTCD | LBTEST | LBORRES | LBORRESU | LBSPEC | LBMETHOD | LBDTC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | ABC | ABC-001 | FreeStyle Libre 3 CGM | 1 | DB14687X-1D25-4A84-8967-xxxxxxxxxxxx 1/27/2024 10:57 | GLUCPE | Plasma Equivalent Glucose | 82 | mg/dL | INTERSTITIAL FLUID | ELECTROCHEMICAL AMPEROMETRY | 2024-01-27T08:10:57 |
| 2 | ABC | ABC-001 | FreeStyle Libre 3 CGM | 2 | DB14687X-1D25-4A84-8967-xxxxxxxxxxxx 1/27/2024 11:02 | GLUCPE | Plasma Equivalent Glucose | 89 | mg/dL | INTERSTITIAL FLUID | ELECTROCHEMICAL AMPEROMETRY | 2024-01-27T11:02 |
| 3 | ABC | ABC-001 | FreeStyle Libre 3 CGM | 3 | DB14687X-1D25-4A84-8967-xxxxxxxxxxxx 1/27/2024 11:07 | GLUCPE | Plasma Equivalent Glucose | 90 | mg/dL | INTERSTITIAL FLUID | ELECTROCHEMICAL AMPEROMETRY | 2024-01-27T11:07 |
| 4 | ABC | ABC-001 | FreeStyle Libre 3 CGM | 4 | DB14687X-1D25-4A84-8967-xxxxxxxxxxxx 1/27/2024 11:08 | GLUCPE | Plasma Equivalent Glucose | 94 | mg/dL | INTERSTITIAL FLUID | ELECTROCHEMICAL AMPEROMETRY | 2024-01-27T11:08 |
DHT Portal CGM Example 2.xlsx